On This Page
Fundamentals of Ionic Equilibrium
Ionic equilibrium represents a specific state in aqueous solutions where molecules and ions coexist in a stable ratio. It primarily involves the balance between un-ionized molecules and the resulting ions produced during the dissolution process in water.
When a solute dissolves in water, it may dissociate into charged particles known as ions. These particles interact with the solvent and each other continuously. The equilibrium state is reached when the dissociation rate equals the recombination rate.
This phenomenon is most prominent in weak electrolytes, which do not fully break apart in solution. Unlike strong electrolytes, weak ones maintain a significant concentration of neutral molecules. This creates a measurable equilibrium constant for the reaction.
Understanding these interactions is vital for predicting the behavior of acids, bases, and salts. It allows chemists to calculate the concentrations of specific species in a mixture. Such calculations are fundamental to pharmacy, biology, and industrial manufacturing processes.
The study of ionic equilibrium requires a firm grasp of thermodynamics and chemical kinetics. It provides the mathematical framework needed to describe how variations in temperature or concentration affect ion levels. This section introduces the core concepts of this balance.
Electrolytes and Ionization
Electrolytes are substances that produce ions when dissolved in polar solvents like water. They are categorized based on their ability to conduct electricity. Strong electrolytes ionize completely, while weak electrolytes only partially dissociate into their constituent ions.
Weak electrolytes are the primary focus of ionic equilibrium studies because they exhibit reversibility. In these systems, the forward reaction of ionization happens simultaneously with the backward reaction of ion pairing. This leads to a dynamic steady state.
The degree of ionization, often denoted by ##\alpha##, measures the fraction of the total electrolyte that exists as ions. For strong electrolytes, ##\alpha## is approximately one. For weak electrolytes, ##\alpha## is significantly less than one.
Factors such as concentration, temperature, and the nature of the solvent influence the degree of ionization. Generally, increasing the dilution of a solution increases the ionization degree. This relationship is central to understanding how electrolytes behave in various environments.
Ionic equilibrium equations are written to show this reversible process clearly. For a general electrolyte AB, the process is represented as ##AB \rightleftharpoons A^+ + B^-##. This visual representation highlights the bidirectional nature of the molecular transformation.
Dynamic Nature of Equilibrium
The term dynamic equilibrium implies that the reaction has not stopped at the molecular level. Instead, the ions are constantly forming and recombining at identical rates. This results in no net change in the macroscopic concentration of the species.
At equilibrium, the chemical potential of the reactants equals the chemical potential of the products. This thermodynamic stability ensures that the system remains unchanged unless external conditions are modified. It is a state of minimum Gibbs free energy.
Le Chatelier's Principle applies directly to ionic equilibrium systems in aqueous solutions. If the concentration of an ion is increased, the equilibrium shifts to consume that excess. This shift helps maintain the balance defined by the equilibrium constant.
The equilibrium constant for ionization is specific to the substance and the temperature. It provides a quantitative measure of the strength of the electrolyte. A higher constant indicates a greater tendency for the molecule to dissociate into ions.
Monitoring these dynamic changes requires sensitive analytical techniques like conductivity measurements. These tools help scientists determine the exact point where equilibrium is established. This data is crucial for designing chemical buffers and specialized reaction environments.
Quantitative Laws and Constants
Quantitative analysis of ionic equilibrium relies on mathematical laws that relate concentration to ionization. These laws allow for the derivation of constants that characterize specific chemical species. They provide a predictable model for complex chemical behavior in solutions.
The Law of Mass Action is the foundation for all equilibrium calculations. it states that the rate of a reaction is proportional to the product of the active masses. In dilute solutions, molar concentration serves as the active mass.
By applying this law, we can derive the equilibrium constant expression for any ionization process. This constant remains unchanged regardless of the initial concentrations used. It is a fundamental property of the electrolyte at a given temperature.
Calculations often involve determining the concentration of hydrogen or hydroxyl ions. These values are essential for defining the acidity or alkalinity of the system. Precise mathematical modeling ensures accuracy in laboratory experiments and industrial processes.
This section examines the specific laws, such as Ostwald’s Dilution Law, that govern weak electrolytes. We will explore how these equations are used to find the degree of dissociation. These tools are indispensable for any student of physical chemistry.
Ostwald’s Dilution Law
Ostwald’s Dilution Law provides a mathematical relationship between the dissociation constant and the degree of ionization. It is specifically applicable to weak electrolytes in dilute solutions. The law demonstrates how dilution affects the ionization process.
For a binary electrolyte with concentration ##C## and degree of dissociation ##\alpha##, the law is expressed as:
This formula links the equilibrium constant ##K## to the concentration and ionization degree.
In cases where the electrolyte is very weak, ##\alpha## is much smaller than one. The equation simplifies to
, allowing for easier calculation. This approximation is standard in most introductory chemistry problems involving weak acids.
From this simplified form, we can derive that ##\alpha = \sqrt{K/C}##. This shows that the degree of ionization is inversely proportional to the square root of the concentration. Consequently, as a solution becomes more dilute, ionization increases.
While the law is powerful, it fails for strong electrolytes because they are already fully ionized. Additionally, it deviates at high concentrations where inter-ionic attractions become significant. It remains a cornerstone for understanding weak acid-base behavior in chemistry.
Ionization Constant of Acids and Bases
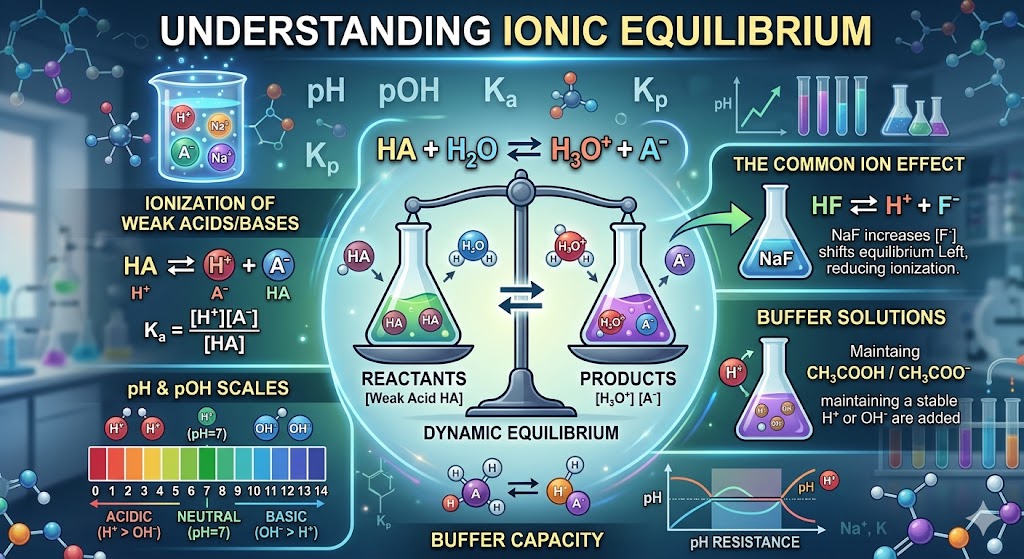
The ionization constant of an acid, denoted as ##K_a##, measures its strength in water. For a weak acid HA, the equilibrium is ##HA \rightleftharpoons H^+ + A^-##. The constant is the ratio of products to reactants.
Similarly, for a weak base BOH, the ionization constant is denoted as ##K_b##. The equilibrium is represented as ##BOH \rightleftharpoons B^+ + OH^-##. A larger ##K_b## value indicates a stronger base with higher hydroxide ion concentration.
The relationship between ##K_a## and ##K_b## for a conjugate acid-base pair is constant. In water at 25 degrees Celsius, their product equals the ionic product of water, ##K_w##. This relationship is essential for interconverting acid and base strengths.
These constants are typically expressed in logarithmic form as ##pK_a## or ##pK_b##. This scale makes it easier to compare substances that vary by several orders of magnitude. Lower ##pK_a## values represent relatively stronger acids within the weak category.
Experimental determination of these constants involves titration or pH measurements. Knowing these values allows chemists to predict the pH of a solution at any concentration. This is vital for controlling chemical reactions in biological and synthetic systems.
pH Scale and Buffer Systems
The pH scale is a logarithmic tool used to express the acidity or basicity of a solution. It simplifies the handling of very small hydrogen ion concentrations. This scale is fundamental to understanding ionic equilibrium in aqueous environments.
A neutral solution has a pH of 7 at room temperature. Values below 7 indicate acidic conditions, while values above 7 indicate basic conditions. This standardized system allows for clear communication of chemical properties across different scientific disciplines.
Buffer systems are specialized solutions that resist changes in pH when small amounts of acid or base are added. They consist of a weak acid and its conjugate base or vice versa. They are essential for maintaining stability.
The ability of a buffer to maintain pH is known as its buffer capacity. This capacity depends on the concentrations of the buffer components. Higher concentrations generally lead to a more robust resistance to pH fluctuations during reactions.
In this section, we will delve into the mathematical definitions of pH and pOH. We will also examine the Henderson-Hasselbalch equation, which is the primary tool for buffer calculations. These concepts are critical for biological and chemical stability.
The Concept of pH and pOH
The term pH is defined as the negative logarithm of the hydrogen ion activity. In most practical applications, molar concentration is used instead of activity. The formula is written as ##pH = -\log[H^+]## for convenience.
Similarly, pOH measures the concentration of hydroxide ions in a solution. It is defined by the equation ##pOH = -\log[OH^-]##. Both scales provide a way to quantify the ionic balance of water-based systems effectively.
The sum of pH and pOH is always constant for a given temperature. At standard room temperature, this sum is exactly 14. This relationship allows for the calculation of one value if the other is known by the chemist.
The logarithmic nature of the scale means that a change of one pH unit represents a tenfold change in ion concentration. This makes the scale highly sensitive to small shifts in chemical composition. It is a powerful tool for monitoring reactions.
Precise pH measurement is achieved using electronic pH meters or chemical indicators. Indicators change color at specific pH ranges, providing a visual cue for equilibrium shifts. Meters provide digital accuracy for sensitive laboratory procedures and industrial monitoring.
Mechanism of Buffer Solutions
Buffer solutions operate through the principle of the common ion effect. When an acid is added to a buffer, the conjugate base reacts with the extra hydrogen ions. This prevents a significant drop in the overall pH level.
Conversely, if a base is added, the weak acid component of the buffer provides hydrogen ions to neutralize the hydroxide. This reaction maintains the hydroxide concentration at a nearly constant level. The system effectively absorbs the external chemical shock.
The pH of a buffer solution can be calculated using the Henderson-Hasselbalch equation. For an acidic buffer, it is:
This equation relates the pH to the acid's strength and component ratio.
Biological systems, such as human blood, rely heavily on buffer systems to function. The carbonic acid-bicarbonate buffer keeps blood pH within a very narrow range. Any significant deviation from this range can be fatal to the organism.
Industrial applications of buffers include fermentation, electroplating, and leather tanning. In these processes, maintaining a specific pH is necessary for enzyme activity or chemical consistency. Buffers ensure that the desired reactions proceed without interference from environmental changes.
Solubility and Precipitation
Ionic equilibrium also governs the solubility of sparingly soluble salts in water. When such a salt is added to water, a saturated solution is eventually formed. At this point, the dissolved ions are in equilibrium with the solid.
The process involves the solid salt breaking into ions and the ions recombining to reform the solid. This state of saturation represents the maximum amount of solute that can dissolve. It is a dynamic balance between two different phases.
Understanding solubility equilibrium is essential for predicting whether a precipitate will form during a reaction. This is a common task in qualitative and quantitative chemical analysis. It helps in separating different ions from a complex mixture.
The solubility of a substance can be influenced by temperature and the presence of other ions. While temperature usually increases solubility, the presence of a common ion typically decreases it. These interactions are mathematically predictable using equilibrium constants.
This final section covers the solubility product constant and the common ion effect. These concepts provide the basis for controlled precipitation in laboratory settings. We will explore the formulas and conditions that determine the fate of dissolved salts.
Solubility Product Constant
The solubility product constant, ##K_{sp}##, is the equilibrium constant for a solid substance dissolving in an aqueous solution. It represents the product of the molar concentrations of the ions raised to their stoichiometric coefficients. It is temperature-dependent.
For a salt like ##AgCl##, the equilibrium is ##AgCl(s) \rightleftharpoons Ag^+(aq) + Cl^-(aq)##. The expression for the solubility product is
. This value is a unique fingerprint for every sparingly soluble compound.
If the product of the ion concentrations in a solution exceeds the ##K_{sp}##, precipitation occurs. This product is called the ionic product, ##Q_{sp}##. Comparing ##Q_{sp}## to ##K_{sp}## tells us if a solution is unsaturated or supersaturated.
Solubility products are used to calculate the molar solubility of a salt in pure water. By setting up an algebraic expression based on the stoichiometry, one can find the concentration of ions. This is a standard calculation in environmental chemistry.
In analytical chemistry, ##K_{sp}## values guide the selective precipitation of ions. By carefully controlling the concentration of a precipitating agent, one ion can be removed while others remain dissolved. This technique is vital for purifying chemical reagents and samples.
Common Ion Effect in Equilibrium
The common ion effect describes the decrease in the solubility of an ionic compound when a salt containing a common ion is added. It is a direct application of Le Chatelier's Principle to ionic equilibrium. The system shifts to restore balance.
For example, adding sodium chloride to a saturated silver chloride solution increases the chloride ion concentration. This causes the equilibrium to shift toward the solid silver chloride. As a result, more silver chloride precipitates out of the solution.
This effect is widely used in the gravimetric analysis of chemical samples. By adding an excess of a common ion, chemists ensure the complete precipitation of the target substance. This improves the accuracy and yield of the chemical measurement.
The common ion effect also plays a role in the ionization of weak acids and bases. Adding a salt of a weak acid suppresses the ionization of that acid. This behavior is the fundamental mechanism behind the operation of buffer solutions.
Understanding this effect is crucial for managing industrial waste and water treatment. By introducing specific ions, harmful heavy metals can be precipitated out of wastewater effectively. This demonstrates the practical importance of mastering the principles of ionic equilibrium.
From our network :
- 10 Physics Numerical Problems with Solutions for IIT JEE
- https://www.themagpost.com/post/analyzing-trump-deportation-numbers-insights-into-the-2026-immigration-crackdown
- Vite 6/7 'Cold Start' Regression in Massive Module Graphs
- EV 2.0: The Solid-State Battery Breakthrough and Global Factory Expansion
- 98% of Global MBA Programs Now Prefer GRE Over GMAT Focus Edition
- Mastering DB2 12.1 Instance Design: A Technical Deep Dive into Modern Database Architecture
- https://www.themagpost.com/post/trump-political-strategy-how-geopolitical-stunts-serve-as-media-diversions
- AI-Powered 'Precision Diagnostic' Replaces Standard GRE Score Reports
- Mastering DB2 LUW v12 Tables: A Comprehensive Technical Guide
RESOURCES
- (Ionic equilibrium) Can someone please explain me how does the ...
- Chemical equilibrium - Wikipedia
- (Ionic equilibrium) This is from my chemistry textbook, can someone ...
- Electrostatic interactions and Donnan equilibrium explain the effects ...
- Chemical equilibrium | Definition, Equation, & Facts - Britannica
- What is ionic equilibrium? - Characteristics | CK-12 Foundation
- CHM 140 - General Chemistry II -
- the ionic product for water: Kw - Chemguide
- Physiology, Resting Potential - StatPearls - NCBI Bookshelf - NIH
- Can you explain the concept of chemical equilibrium and give an ...
- Physiology, Action Potential - StatPearls - NCBI Bookshelf - NIH
- A Clear Explanation on The Key Concept of Ionic Equilibrium
- The membrane potential - Khan Academy
- Chemical Equilibrium - BYJU'S
- Resting Membrane Potential - Nernst - TeachMe Physiology
11 Comments