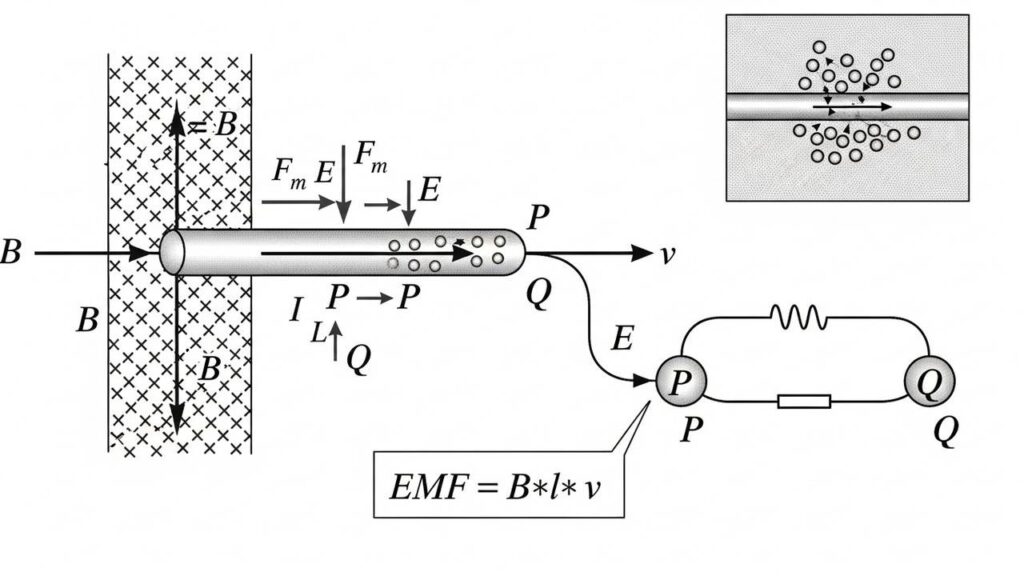
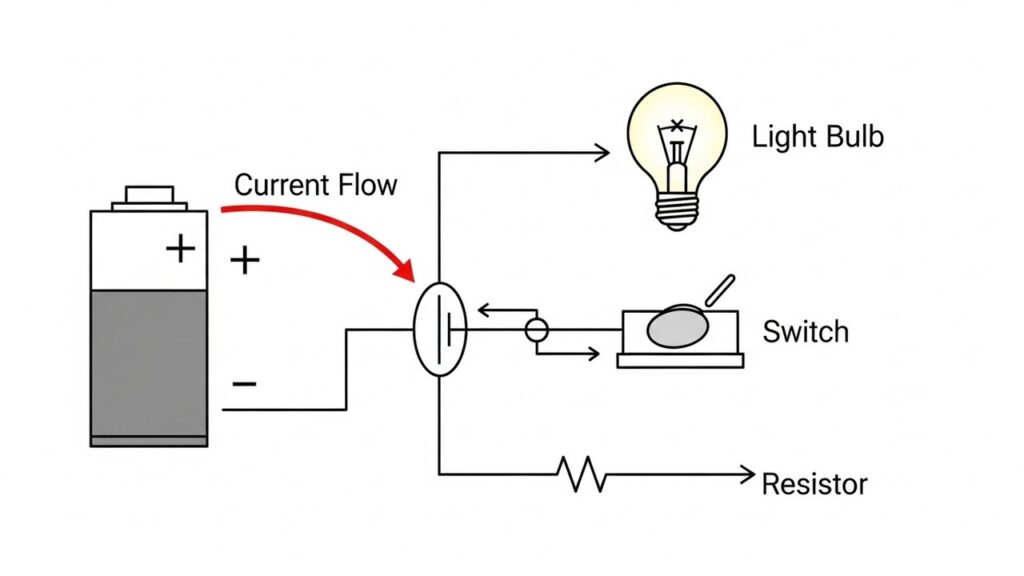
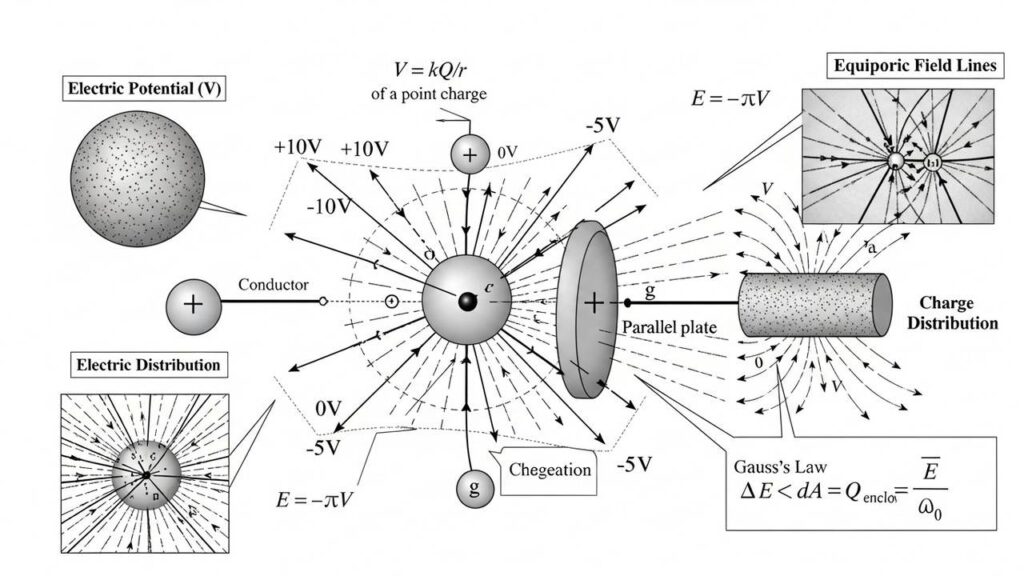
VIVA QUESTION
Resources & Insights
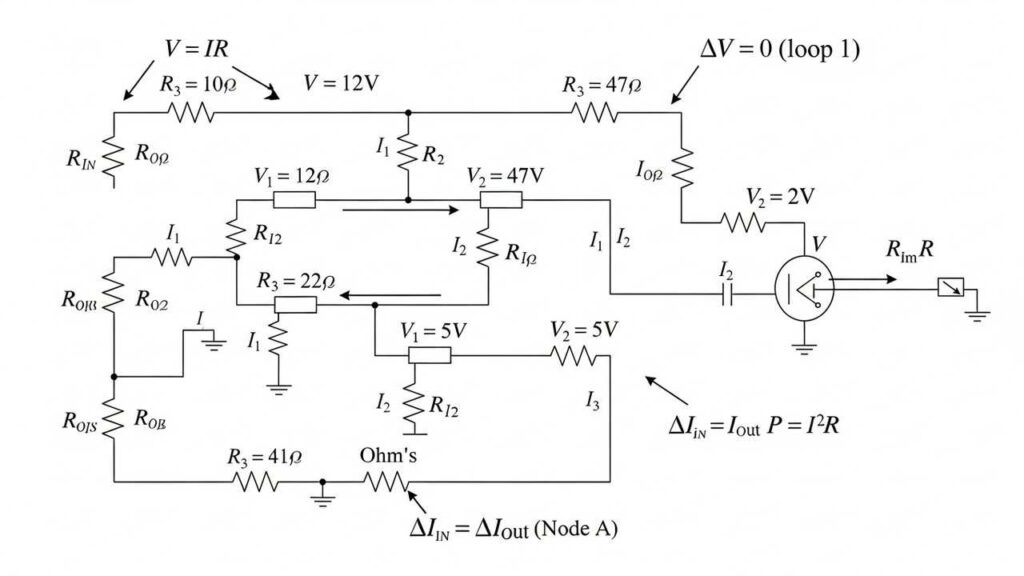
Problems on resistance, circuits, and current distribution.
Strengthen circuit analysis. READ MORE...

Momentum Mastery Quiz: From Fundamentals to Advanced Concepts
This Momentum Mastery Quiz is designed to assess and enhance your comprehensive understanding of momentum in physics. Featuring 25 multiple-choice questions, the quiz covers a wide range of topics from basic definitions and calculations of linear momentum to more advanced concepts such as impulse, conservation laws, elastic and inelastic collisions, and relativistic momentum. Each question is thoughtfully crafted to challenge your knowledge and application of momentum principles. Ideal for students, educators, and physics enthusiasts, this quiz serves as an effective tool to reinforce learning, identify areas for improvement, and deepen your grasp of momentum-related theories and real-world applications. Engage with these questions to test your proficiency and advance your mastery of momentum. READ MORE...

Navigating the CBSE Board Exams 2025: A Comprehensive Guide
Conquer the CBSE Board Exams 2025 with our guide! Learn effective study strategies, time management tips, and overcome exam anxiety for success. READ MORE...

Metal compound A reacts with dilute hydrochloric acid to produce effervescence. The gas evolved extinguishes a burning candle. Write a balanced chemical equation for the reaction if one of the compounds formed is calcium chloride. Also, determine the Metal Compound A.
Based on the information provided, the metal compound A reacts with dilute hydrochloric acid to produce effervescence, and the gas evolved extinguishes a burning candle. Additionally, one of the compounds formed in the reaction is calcium chloride. Let's write the balanced chemical equation for this reaction: Metal Compound A + Hydrochloric Acid → Calcium Chloride + Gas + Water Since calcium chloride is formed, we can represent it as CaCl2. The gas evolved that extinguishes a burning candle is hydrogen gas (H2). The balanced chemical equation for the reaction is as follows: Metal Compound A + 2HCl → CaCl2 + H2 + H2O In this reaction, the metal compound A reacts with hydrochloric acid (HCl) to produce calcium chloride (CaCl2) […] READ MORE...
Which gas is usually liberated when an acid reacts with a metal? Illustrate with an example. How will you test for the presence of this gas?
When an acid reacts with a metal, hydrogen gas (H2) is usually liberated. The reaction between an acid and a metal is a type of single-displacement or single-replacement reaction, where the more reactive metal displaces hydrogen from the acid, forming a metal salt and releasing hydrogen gas. Here's an example of the reaction between hydrochloric acid (HCl) and zinc (Zn): Hydrochloric Acid + Zinc → Zinc Chloride + Hydrogen Gas 2HCl + Zn → ZnCl2 + H2 Testing for the Presence of Hydrogen Gas: To test for the presence of hydrogen gas, you can perform the "pop" test. This test is a simple way to detect the presence of hydrogen gas, as hydrogen is highly flammable. When hydrogen gas is […] READ MORE...