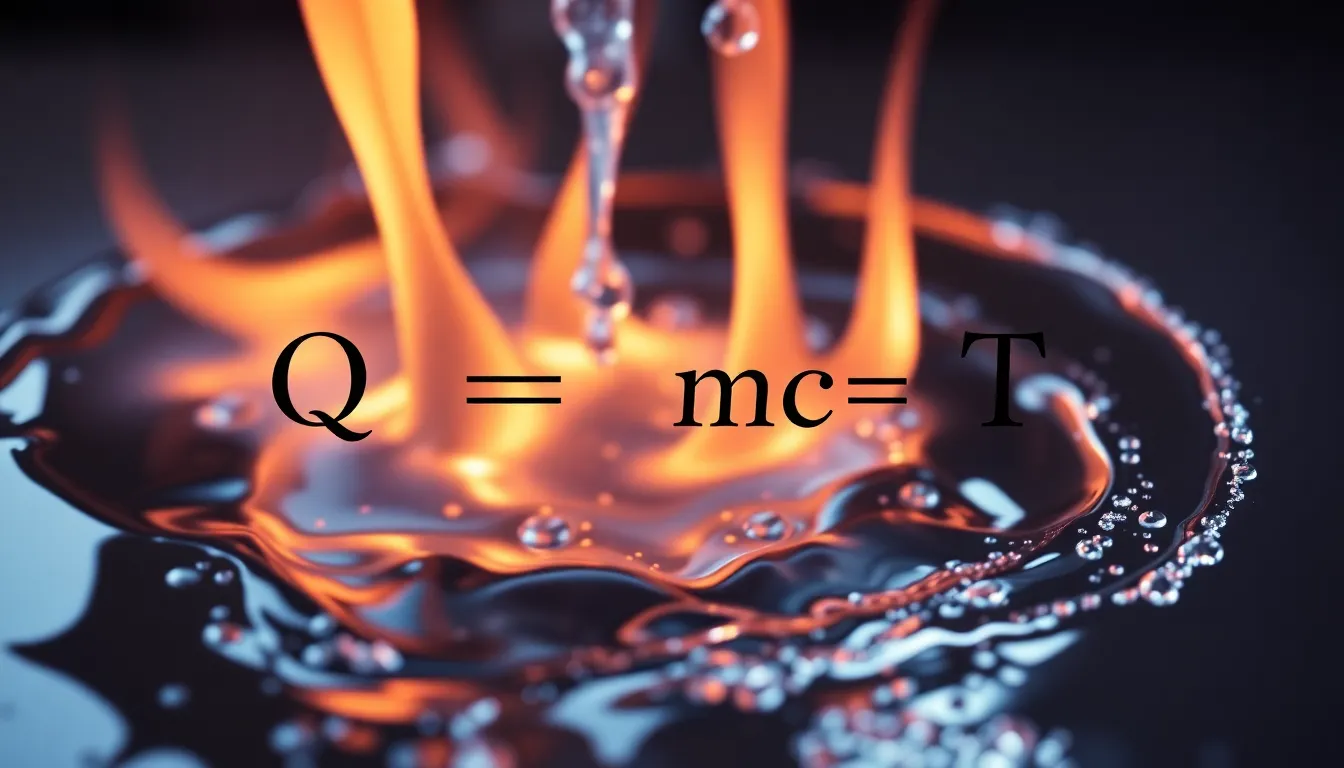Where Exploration Leads to Academic Excellence
A Knowledge Hub by Rahul Anand

Mathematical Marvels: Insights from the Field
Systems of Linear Equations: Solving Solving ##3x+2y=16 \text{ and } x-y=2##
A technical guide to solving systems of linear equations using substitution and matrix methods, including detailed proofs and historical context.
Solving Quadratic Equations ##x^2 – 7x + 10 = 0##
A professional deep dive into solving quadratic equations using the factoring method. Step-by-step analysis of ##x^2 – 7x + 10 = 0## with technical proofs and historical context.
Simplify ## \frac{x^2 – 9}{x^2 + 5x + 6} ##
Learn how to simplify ##\frac{x^2 – 9}{x^2 + 5x + 6}## using difference of squares and trinomial factoring while identifying excluded values and discontinuities.
Simplifying Polynomial Expressions: Technical Step-by-Step Guide
A technical overview of simplifying polynomial expressions, focusing on distribution, combining like terms, and verification through numerical and calculus-based methods.
Linear Equation with One Variable: Technical Solution and Analysis
A technical guide to solving the linear equation with one variable 5x – 12 = 3x + 8. Includes step-by-step algebraic reduction, verification, and historical context.
SASTRA Ramanujan Award 2025: Breakthroughs in Number Theory
Learn about SASTRA Ramanujan Award 2025 winner Dr. Alexander Smith and his work on congruent number problems and class group distributions.
National Mathematics Day 2025: Ramanujan’s Influence on AI
National Mathematics Day 2025 celebrates the profound legacy of Srinivasa Ramanujan and his impact on modern AI and physics.
NEP 2020 math curriculum: Integrating AI and Ancient Heritage
Discover how the NEP 2020 math curriculum in 2025 is revolutionizing Indian classrooms by combining AI literacy with ancient mathematical heritage.
Calculus Castle
Understanding L'Hôpital's Rule for Limits: A Comprehensive Guide
Learn how L'Hôpital's rule helps evaluate indeterminate forms in limits. This guide provides a complete explanation and examples.
Understanding Implicit Differentiation and Differential Equations
Explore implicit differentiation and its relation to differential equations in our new quiz feedback.
Understanding Sophie Germain Primes: Properties Applications and Algorithms
Discover Sophie Germain primes special prime numbers with unique properties. Learn about their relation to safe primes applications in cryptography and how to identify them using Python.
Proving the Generalized Function Equality for Cosine Series
Prove the equality of cosine series in generalized functions. Learn how to manipulate trigonometric series for advanced applications.
Finding the Value of ‘a’ for Piecewise Function Continuity at x = 0
Determine the value of ‘a’ to ensure continuity of a piecewise function at x = 0 using limits and Taylor series.
Finding the Limit of a Trigonometric Function: Limit of Cosine to the Power of x
Calculate the limit of cos(√x) raised to the power of 1/x as x approaches 0 from the right. Learn the Taylor expansion method for solving this type of limit problem.
Evaluating the Exponential and Logarithmic Limit: x^x as x approaches 0+
Learn how to evaluate the limit of x^x as x approaches 0 from the positive side using logarithms and L’Hôpital’s rule. Find the answer!
Finding the Limit Using L’Hôpital’s Rule: x ln(1 + 1/x)
Learn how to evaluate the limit of x times the natural log of (1 + 1/x) as x approaches infinity using L’Hôpital’s Rule.
Evaluating the Trigonometric Limit: lim x→0 (sin(5x) – sin(3x))/x^3
Find the limit of a trigonometric expression as x approaches 0. Learn how to use trigonometric identities to solve this problem.
Understanding Vectors in Mathematics: Definition Operations and Applications
Learn about vectors in mathematics their properties and how they’re used in physics computer graphics and machine learning.
Insight into the Laws of Physics
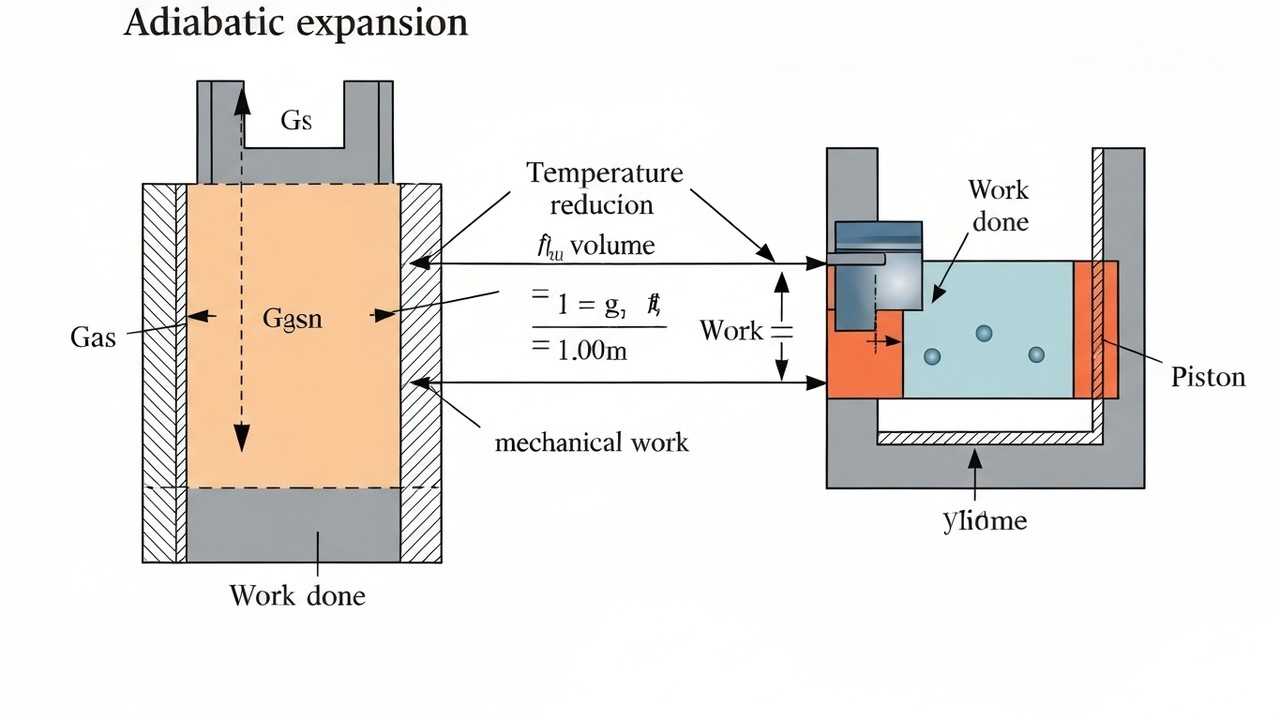
Adiabatic Expansion Work: Calculating Energy Changes without Heat Transfer
Understand adiabatic expansion work, solving a problem to calculate the work done by a gas during an expansion without heat exchange.
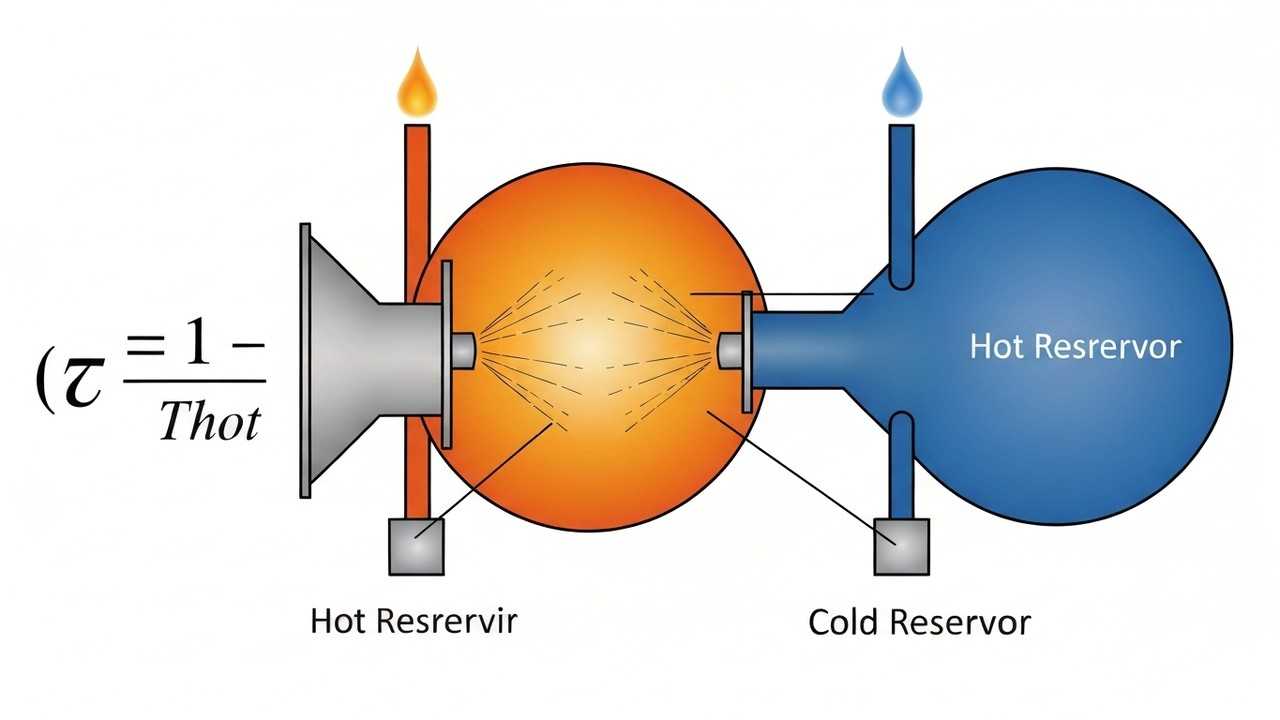
Carnot Efficiency Calculation: Ideal Heat Engine Performance and Limitations
Learn Carnot efficiency calculation, explaining the theoretical limits of heat engine performance and solving a related problem.
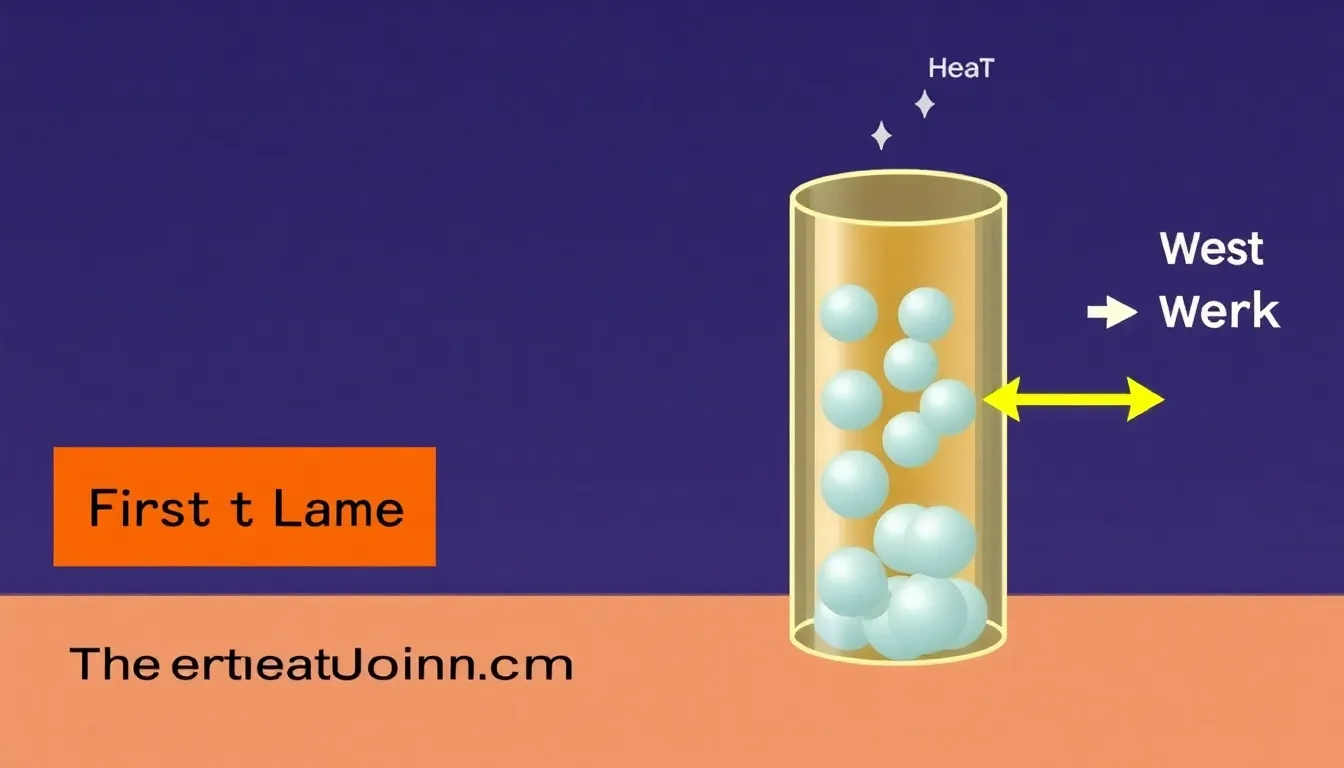
First Law Thermodynamics Problem: Understanding Energy Conservation in Systems
Tackle a first law thermodynamics problem, explaining how internal energy, heat, and work interrelate in a system.
Stefan-Boltzmann Radiation Problem: Calculating Thermal Energy Emission from Surfaces
Understand the Stefan-Boltzmann Law and solve a problem to calculate the radiant power emitted by a black body at a given temperature.
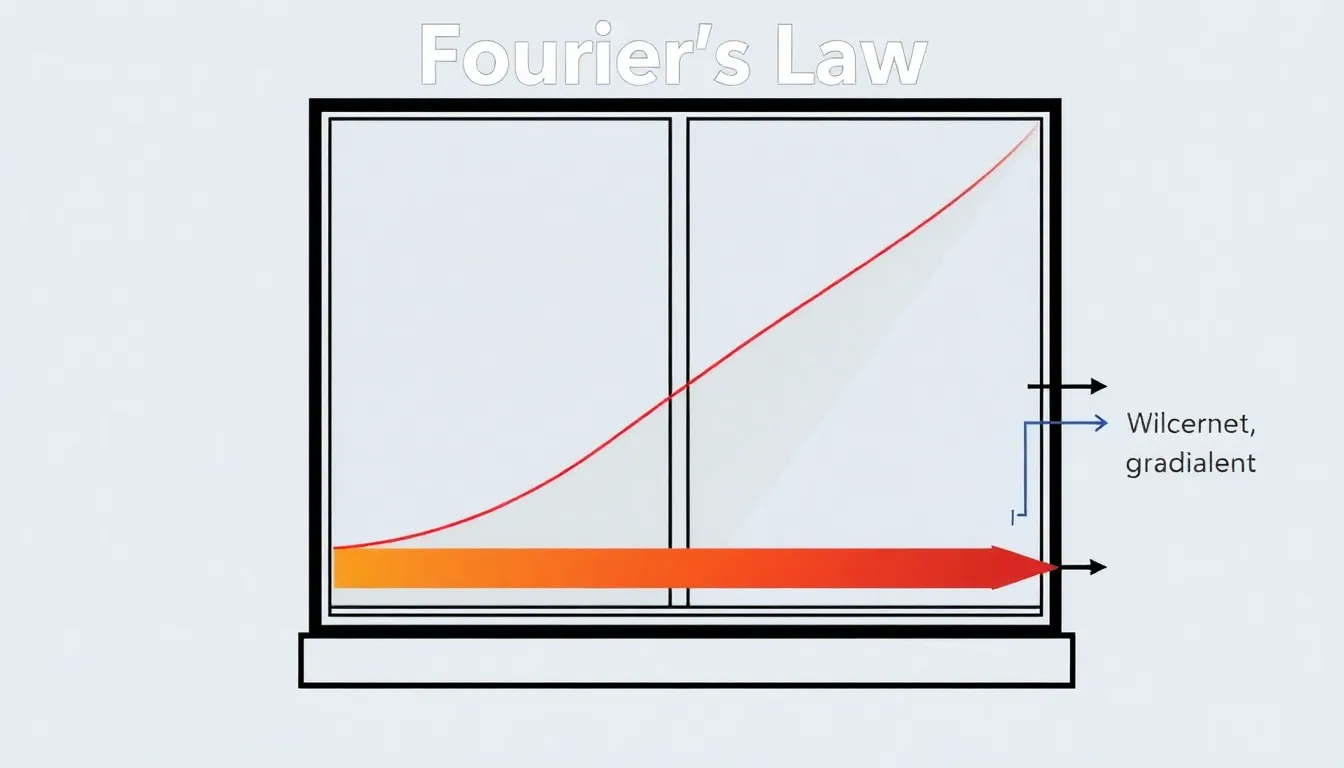
Heat Conduction Rate Calculation: Thermal Flow Through Material Barriers
Learn heat conduction rate calculation using Fourier’s Law, solving a problem to determine heat loss through a windowpane.
Linear Thermal Expansion Example: Calculating Length Changes in Materials with Temperature
Explore linear thermal expansion with a practical example, calculating the change in length for a metal rod due to temperature fluctuation.
Latent Heat Fusion Problem: quantifying energy for phase changes from solid to liquid
Understand the concept of latent heat of fusion and solve a numerical problem calculating the energy needed to melt ice without temperature change.
Calculate Specific Heat Capacity: How Materials Absorb Heat and Change Temperature
Discover how to calculate specific heat capacity, explaining how materials absorb heat, and solve a problem involving energy needed to raise water temperature.
IIT JEE Spring Numericals
IIT JEE Mechanics Numericals
Find all meet instants that match vertical heights with gravity
Find the coasting interval to plan multiphase travel efficiently
Compute time and acceleration by integrating position dependent velocity v(x)
When do they meet? Equations to analyze relative motion precisely
Find arrival time by calculus to solve 1D kinematics accurately
IIT JEE Optics Numericals
NUMERICAL SOLUTIONS IN PHYSICS
Electrostatics Numericals for IIT JEE
Excel in electrostatics numericals.
Elastic Potential Energy Numericals for IIT JEE
Calculate energy in elastic deformations.
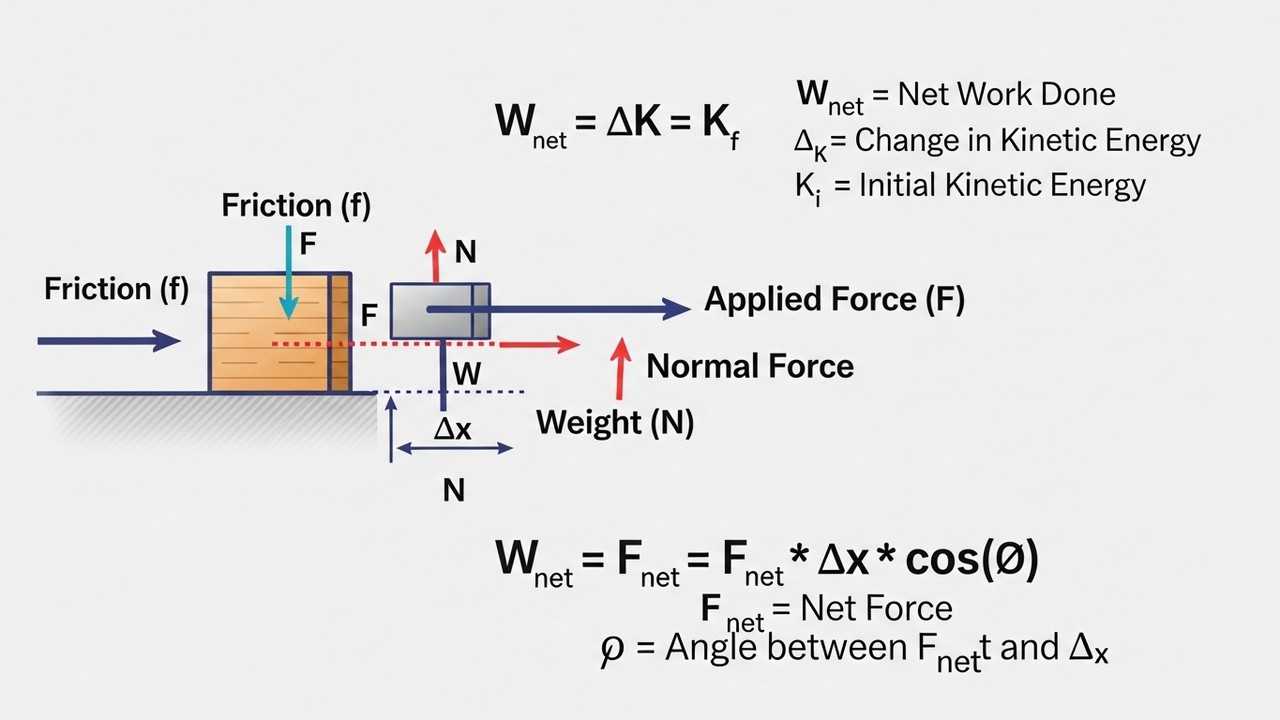
Work and Energy Numericals for IIT JEE
Strengthen energy-based problem solving.
Rotational Motion Numericals for IIT JEE
Boost rotational mechanics problem solving.
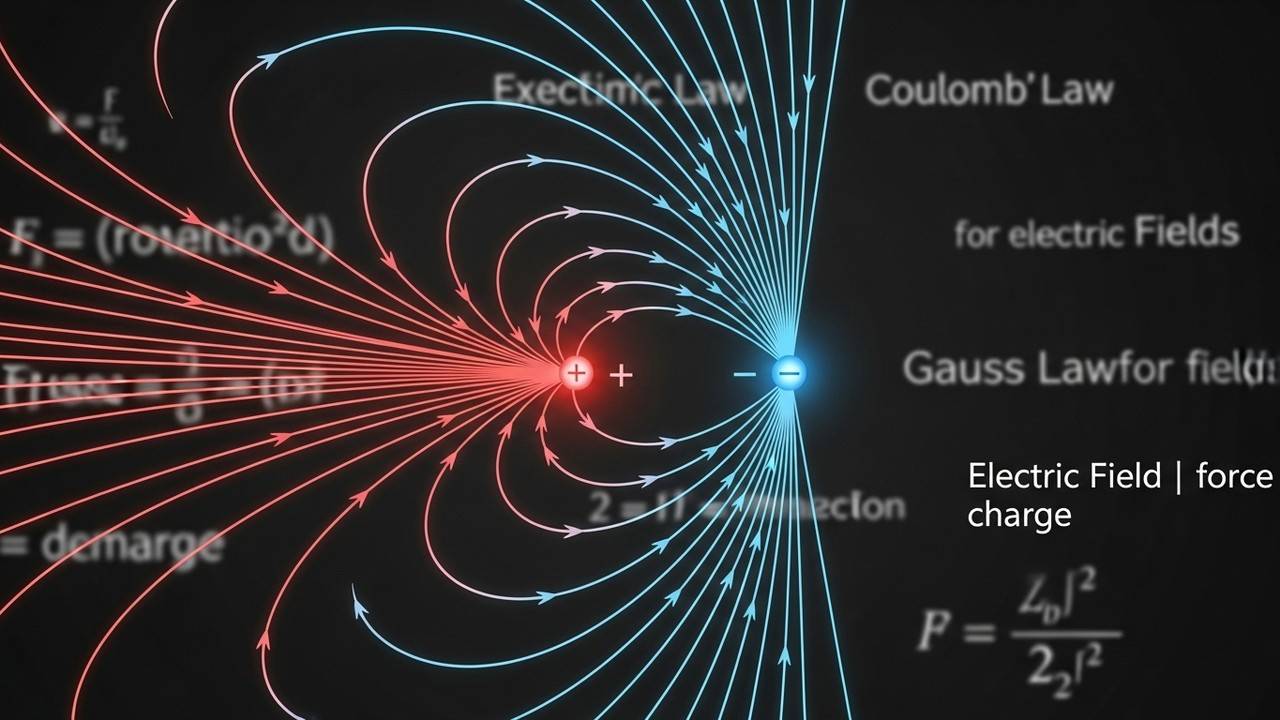
What is Coulomb’s Law and How Did Coulomb Discover It?
Learn about Coulomb’s Law the inverse square law of force and how French physicist Charles-Augustin de Coulomb discovered it.
Understanding Static Electricity: Causes Examples and Prevention
Learn about static electricity from everyday sparks to lightning. Discover causes examples like shocks from metal and how to prevent it.
Calculating Car Distance Traveled with Uniform Acceleration in 10 Seconds
Learn how to calculate the distance a car travels with uniform acceleration. Find the distance covered in 10 seconds.
Understanding Momentum: 10 Examples from Basic to Advanced Physics
Learn about momentum in physics with 10 examples from basic to advanced. Explore how momentum affects objects in motion.
Atom to Atom: Insights from the Chemical Universe
Navigating the CBSE Board Exams 2025: A Comprehensive Guide
Conquer the CBSE Board Exams 2025 with our guide! Learn effective study strategies, time management tips, and overcome exam anxiety for success.
An Introduction to the Periodic Table
The periodic table is a fundamental tool in chemistry, organizing the elements based on their atomic structure and properties. This systematic arrangement not only facilitates the study of chemical behavior but also provides insights into the relationships between…
Acids, Bases and Salts: Comprehensive Guide
Acids, Bases and Salts represent fundamental categories in chemistry that play crucial roles in various chemical reactions and real-world applications. Understanding their properties, behaviour in different reactions, and how they interact with each other forms the…
The Legacy of Puddled Iron in Contemporary Metallurgy
Puddled iron is a refined form of iron produced through a process known as puddling. It was a significant technological innovation during the early stages of the Industrial Revolution, providing a method to transform crude pig iron into a more malleable and durable mate…
What are acids. bases and salts?
These three types of compounds play crucial roles in various chemical reactions and are important in fields such as chemistry, biology, and industry. Understanding their properties and behavior is fundamental in chemistry and helps explain a wide range of natural and ch…
Metal compound A reacts with dilute hydrochloric acid to produce effervescence. The gas evolved extinguishes a burning candle. Write a balanced chemical equation for the reaction if one of the compounds formed is calcium chloride. Also, determine the Metal Compound A.
Based on the information provided, the metal compound A reacts with dilute hydrochloric acid to produce effervescence, and the gas evolved extinguishes a burning candle. Additionally, one of the compounds formed in the reaction is calcium chloride. Let’s write the…
Which gas is usually liberated when an acid reacts with a metal? Illustrate with an example. How will you test for the presence of this gas?
When an acid reacts with a metal, hydrogen gas (H2) is usually liberated. The reaction between an acid and a metal is a type of single-displacement or single-replacement reaction, where the more reactive metal displaces hydrogen from the acid, forming a metal salt and…
Why should curd and sour substances not be kept in brass and copper vessels?
Curd and sour substances should not be kept in brass and copper vessels because these metals can react with acidic foods, leading to potential health hazards. The main concern is the leaching of toxic metals into the food or liquid being stored, which can contaminate…
Life’s Mysteries Revealed: Exploring Biology
AI-generated genomes: Designing the First Synthetic Virus Killers
Learn about the landmark 2025 achievement of AI-generated genomes and how synthetic bacteriophages are being designed to combat antibiotic resistance.
social microbiome: How Social Genetics Shape Your Gut Health
Learn about the social microbiome and how a Dec 23, 2025 study proves your gut bacteria is shaped by the genetics of people you live with.
metal nanoparticles: Precision Stress and the Future of Cancer Shutdown
Learn about how metal nanoparticles are being used to trigger selective oxidative stress in cancer cells, according to breakthrough research released on December 24, 2025.
Personalized CRISPR and Record-Breaking DNA Sequencing: The Dawn of N-of-1
Learn about Personalized CRISPR and Record-Breaking DNA Sequencing and how the convergence of ultra-fast genomics and custom gene editing is revolutionizing medicine in 2025.
MASTERING PLANT BIOLOGY FOR CBSE & NEET (MEDICAL): PATH TO SECURE TOP RANK
Plant Biology is one of the most scoring areas in CBSE Class 11–12 Biology and the NEET (UG) medical entrance exam. Many students ignore it because they “like human physiology more”, but toppers know a secret: 👉 Plant topics are highly repetitive, conceptual, and…
How to Prevent Measles Spread: Canada’s Elimination Status and the Path Forward
A concerning development: Canada has officially lost its measles elimination status, signaling a serious threat that demands immediate attention. This piece dissects the reasons behind this reversal and provides crucial insights into combating the escalating spread of t…
The Legacy of James D. Watson: A Titan of Genetics
You’ll learn to understand DNA structure, a pivotal discovery by James D. Watson and Francis Crick that revolutionized biology and medicine.
Understand Brain Cell Development: Unlocking the Secrets of the Mind
Scientists are mapping brain cell development to understand the brain. You’ll understand brain cell development, which could lead to new treatments.